lake facts
What is a Lake?
It’s not as simple as it may sound. A lake is a body of water but it is so much more. A lake is an ecosystem, a community of interaction among animals, plants, microorganisms, and the physical and chemical environment in which they live. Critical to any lake ecosystem is the lake’s watershed, the surrounding land area that drains into the particular lake. A complex interdependence has evolved among the organisms in a lake community. If one part of the ecosystem is disturbed, it affects other parts. Well balanced ecosystems, however, do change from season to season and from year to year. Short term events, such as an unusual or excessive algal bloom, may not necessarily signal a long-term problem. On the other hand, changes in land use in the watershed may not immediately have a visible effect on the lake. It may take a decade or more for the lake problems to become apparent.
Most lakes were created by geological events. The vast lake-dotted and marshy landscapes found in North America were formed glacier action 10,000 to 20,000 years ago. Glaciers formed lake basins by gouging holes in loose soil or bedrock, by depositing material across stream beds, or by leaving buried chunks of ice whose melting shaped lake basins. More recently, humans and other animals have created lakes and reservoirs by damming rivers and streams.
Lakes constantly undergo evolutionary change, reflecting the changes that occur in their watersheds. Most are destined to fill in with remains of lake organisms and with silt and soil washed in by floods and streams. These gradual changes in the physical and chemical components of a lake affect the development and succession of plant and animal communities.
This natural process by which lakes form, evolve, and disappear takes thousands of years. Human activities, however, can change these lakes for better or worse in less than a single generation.
How Do Lakes Work?
Before deciding how to protect a lake it is important to develop a basic understanding of the physical. biological, and chemical properties of a lake. These properties such as light, temperature, wind, precipitation and nutrients affect plants, animals and the lake itself.
1. A Physical Look at Lakes
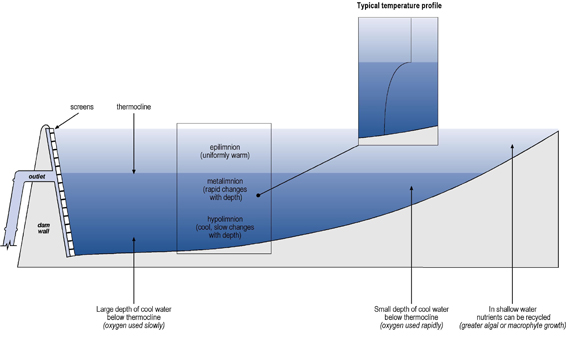
Lakes in temperate climates tend to stratify or form layers, especially during summer, because the density of water changes as it’s temperature changes. Water is most dense at 39 deg.F. Both above and below that temperature, water expands and becomes less dense. This means that in the spring, just before the ice melts, the water near the lake bottom will be at 39 deg.F. Water above the lake bottom will be cooler, approaching 32 deg. Just under the ice. As the weather warms, the ice melts and the surface waters begin to heat up. Wind action and increasing water density causes this surface water to sink and mix with the deeper, a process called spring turnover.
As summer progresses, the temperature and density differences between the upper and lower lake water becomes more distinct, and most deep lakes form three separate layers. The upper layer, the epilimnion, is characterized by warmer water. The epilimnion is roughly equivalent to the zone of light penetration, where the bulk of productivity, or growth occurs. Much of the plant and fish life is found in this zone.
Below the epilimnion is another layer, the thermocline, in which the temperature declines rapidly. The thermocline is a narrow transitional band of transition which helps to prevent the mixing between the layers.
Below the thermocline lies water much colder than the epilimnion, called the hypolimnion. The hypolimnion is the zone of decomposition, where plant material either decays or sinks to the bottom and accumulates. Dissolved oxygen levels are often very low in this layer.
These temperature conditions will continue until fall. Then surface waters cool until they are as dense as the bottom waters and the wind action mixes the lake. This is the fall turnover.
2. A Biological Look at Lakes
A lake can be divided into zones, or communities, of plants and animals. Extending from the shoreline is the littoral community, where aquatic plants are dominant. The size of this community depends on the extent of shallow areas around the lake and the clarity of the water for light penetration. These plants play an important role in the overall aquatic community by producing oxygen and providing food and shelter for insects, crustaceans, frogs, turtles and fish. Maintaining the health and integrity of this zone is critical to the overall health of the lake.
The area of open water is the limnetic community. This area is the habitat of phytoplankton (algae),zooplankton (microscopic animals), and fish. Phytoplankton are very important, serving as the base of the lake’s food chain and producing oxygen.
The process by which green plants, including algae, produce oxygen from sunlight, water, and carbon dioxide is photosynthesis. Chlorophyll is a pigment produced by the plants, which is essential for this process. Since sunlight is very important for photosynthesis, oxygen will be produced only as deep as the sunlight penetrates. The depth of light penetration can be measured using a Secchi disk.

Below the limnetic zone is the profundal community, where light does not penetrate. This zone or community is dominated by respiration, or oxygen consumption, rather than oxygen production. This zone corresponds roughly to the hypolimnion layer. The community in this zone consists of such organisms as bacteria and fungi. These organisms decompose dead plants and animals that descend from the waters above. This process consumes oxygen.
The presence or absence of oxygen in the different water zones determines where organisms such as fish and zooplankton are found. In spring, when the lake water is well mixed, oxygen is usually present at all depths and organisms may be distributed throughout the lake. In the summer, under stratified conditions, little or no oxygen is produced in the hypolimnion. Available oxygen is consumed through decomposition of plant and animal material, and oxygen levels become too low for fish who must move to the top layer, or epilimnion.
If these conditions are prolonged and the upper layers become too warm, cold-water fish such as trout may become stressed and eventually die. In the fall, the lake layers break down and turnover replenishes oxygen to the bottom waters.
The formation of ice in winter severs the atmospheric supply of oxygen to the lake. If sunlight can penetrate, plants continue to produce oxygen but if it doesn’t the amount of oxygen consumed may exceed the amount produced. This is quite common in lakes that have large amounts of weeds, leaves and other organic debris. If oxygen levels fall too low, fish and other aquatic life may die, with game fish such as walleye and bass often being among the first to succumb.
3. A Chemical Look at Lakes
Plants require various substances for growth, including phosphorus, carbon, and nitrogen. The concentrations of these substances in water control the total amount of plant material that can grow. The quantity of each required substance varies. For example, a high percentage of all plant matter is carbon and a very small percentage is phosphorus. If any one of these substances is absent, plants cannot grow, even if the other substances are abundantly available.
In many lakes, phosphorus is the least available nutrient, therefore it’s quantity controls the extent of algal growth. If more phosphorus is added to the lake from lawn fertilizers, septic tank runoff, or even from phosphorus rich sediments stirred up from the lake bottom, more algae will grow.
In turn, the amount of algae in the water will determine how deep light penetrates as measured by the Secchi disk. Combined measurements of phosphorus level, algae abundance(expressed in terms of chlorophyll) and Secchi disk transparency are used to identify the tropic status or the level of growth of a lake.
A eutropic or nutrient-rich lake tends to be shallow,”green”,and has limited oxygen in the hypolimnion. An oligotropic lake is relatively nutrient poor, is clear and deep, and has a hypolimnion high in dissolved oxygen. A mesotropic lake is intermediate between the two. Factors vary, however, from lake to lake, and assessments are necessarily subjective.
Other chemical factors also play an important role in lake ecology. The acidity of water, measured by the PH scale, is an important consideration for aquatic life. A desirable range in PH for aquatic life is 6.5 to 9.0. Values either higher or lower may interfere with reproduction, respiration, and other biological functions of aquatic life. Alkalinity, or buffering capacity, determines the ability of water to withstand great fluctuations in PH. The alkalinity of a lake generally depends on minerals, such as lime in it’s watershed. Watersheds with soils rich in lime and related materials will provide much buffering to lakes, while those poor in lime provide very little buffering and are more susceptible to changes in PH and acid deposition or acid runoff.
There’s more to a lake than meets th eye! The physical, biological and chemical factors that influence the workings of a lake provide the foundation needed to understand what can go wrong in a lake , and what to do about it.
![Lake Wononscopomuc Association [logo]](http://lakewononscopomuc.com/charon/wp-content/themes/lake-association/img/Logo100.png)




